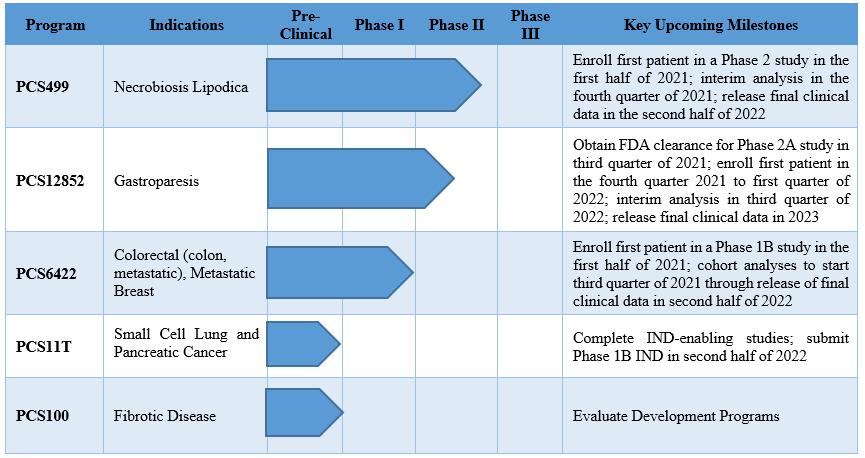
Applied StemCell Inc on X: "ASC can develop preclinical assays to determine efficacy, pharmacodynamic, dose-ranging studies, biodistribution, and develop other IND-enabling assays to measure potency and toxicity of the drug including NHPs,

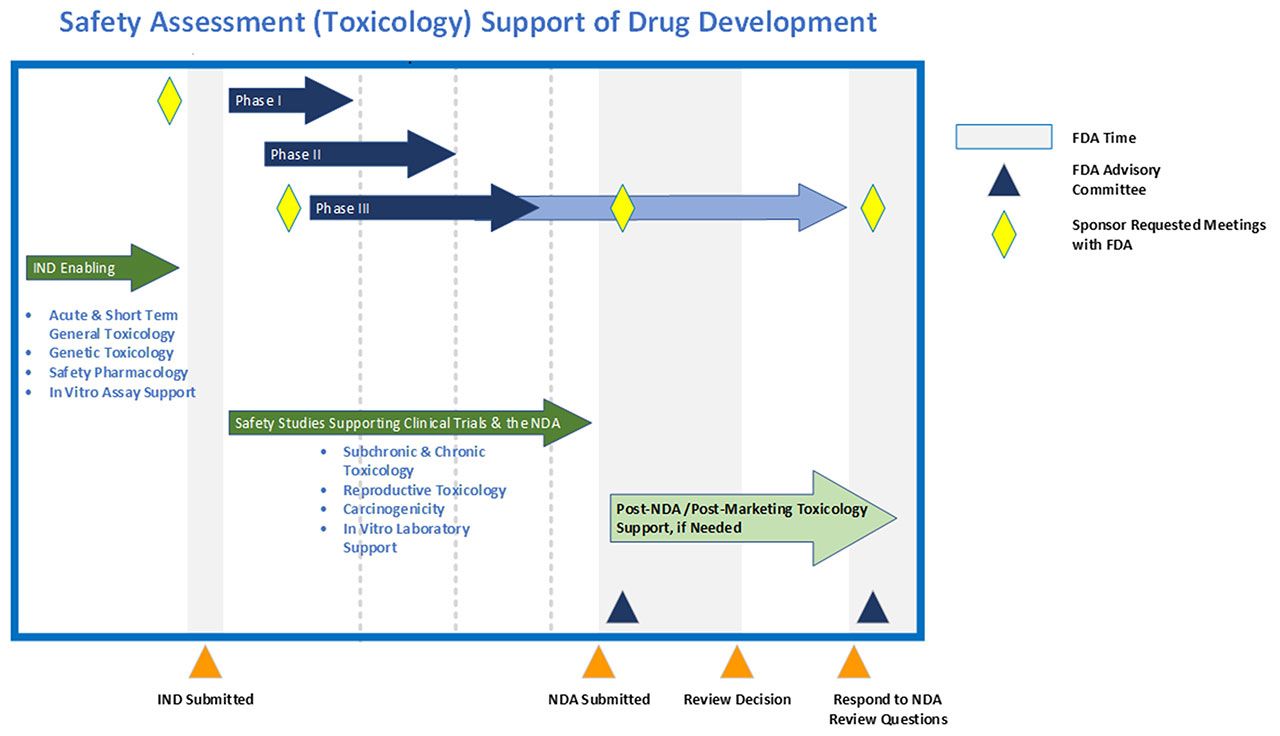
The alignment of major PK/PD related studies with the decision points... | Download Scientific Diagram
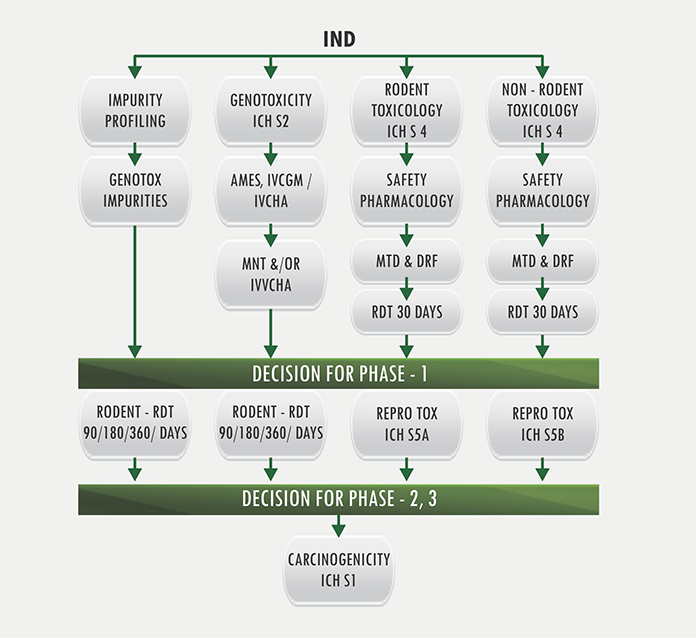
Preclinical pharmacology in IND-enabling studies and clinical pharmacology in clinical protocol development